|
11/16/2023 0 Comments Oxygen valence electrons 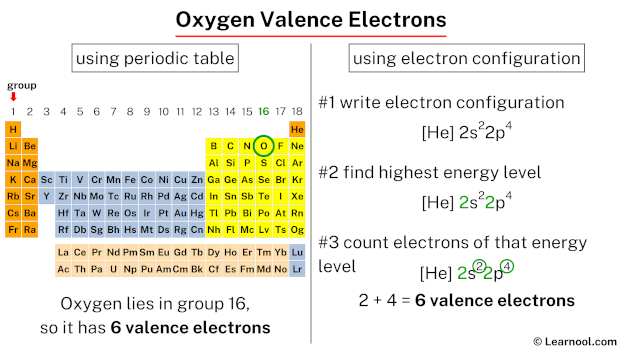
Here no doubt electrons fill in penultimate shell but there is only one electron in valence shell that is 4s. The farthest shell from the nucleus is 2. The electronic configuration of O is 1s2 2s2 2p4. 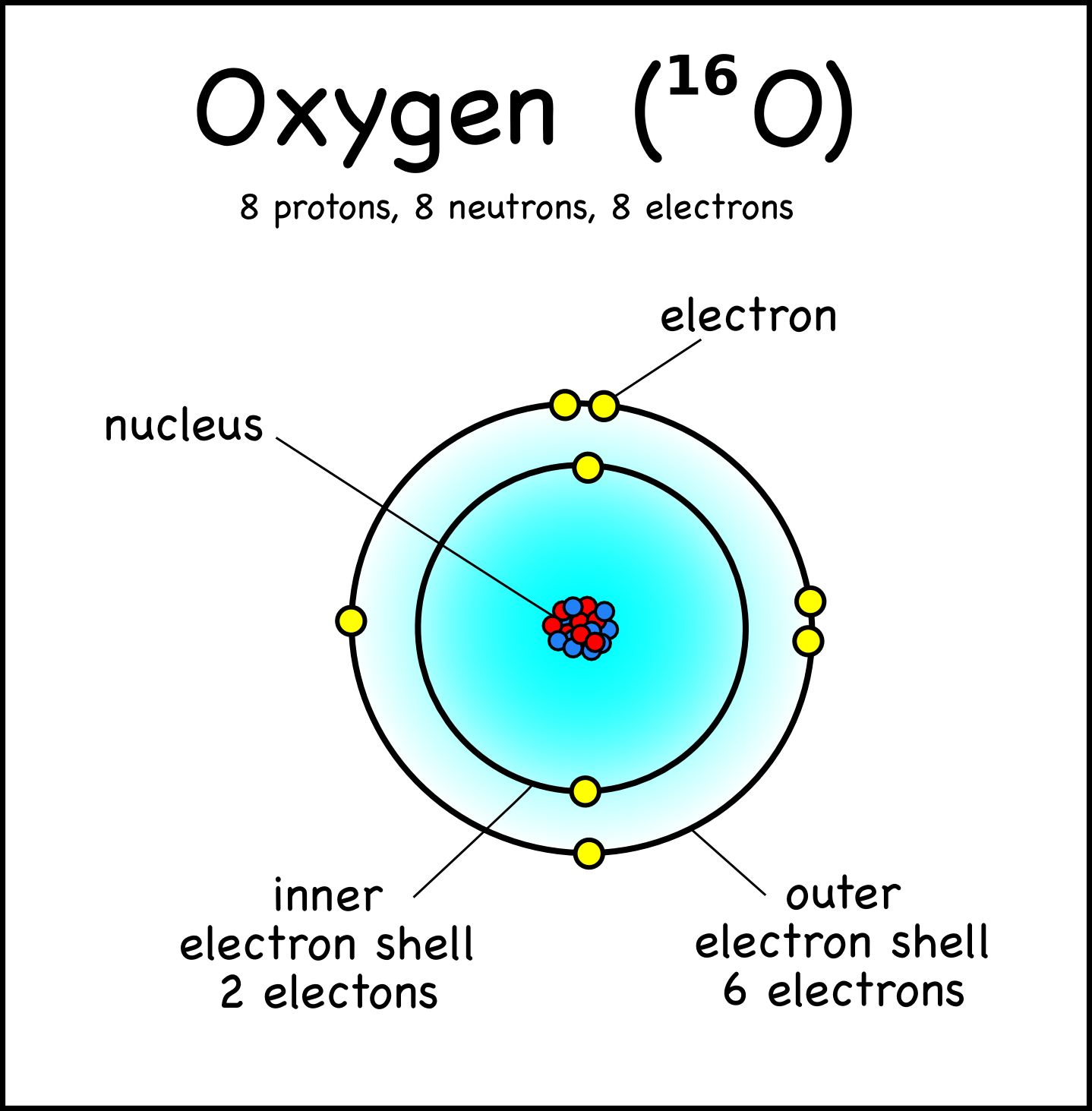
The atomic number of copper is 29 with electronic configuration 1s2 2s2 p6 3s2 p6 d10 4s1 ). The Atomic Number(Z) of Oxygen (O) is 8, corelating to 8 electrons. So, O4 would mean four oxygen atoms, and its molar mass would be 16 4. It is more complex to determine the number of valence electrons in some elements like copper. The premise of VSEPR is that the valence electron pairs surrounding an atom. Similarly there are seven valence electrons in bromine (Br = 1s22s2p63s2p6d104s2p5). Hence 3s is the valance shell and electrons in this shell are valence electrons of Mg. There are three shells, out of that third shell consist of two electrons. For example, Atomic number of magnesium is 12, therefore electronic configuration will be 1s22s2p63s2. Therefore valence electrons are responsible for different chemical and physical properties of an element. Each oxygen atom has 6 valence electrons in its outer energy level When two oxygen atoms get close to each other, the attractions from the nucleus of both. The outermost energy level in an atom is called as valence shell and electrons placed in this shell are known as valence electrons. These electrons are mainly in the bond formation and chemical reaction with other atoms. 
First electrons filled in low energy levels and then move to higher energy level. In an atom, electrons filled in different levels according to their energies. Valence Electrons Configuration and the Periodic Table 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
